CDC recommends updated Covid vaccines for everyone ages 6 months and up, allowing shots to start within days
CDC Director Mandy Cohen still has to sign off on the panel's recommendation before the new Pfizer and Moderna Covid vaccines become available nationwide.
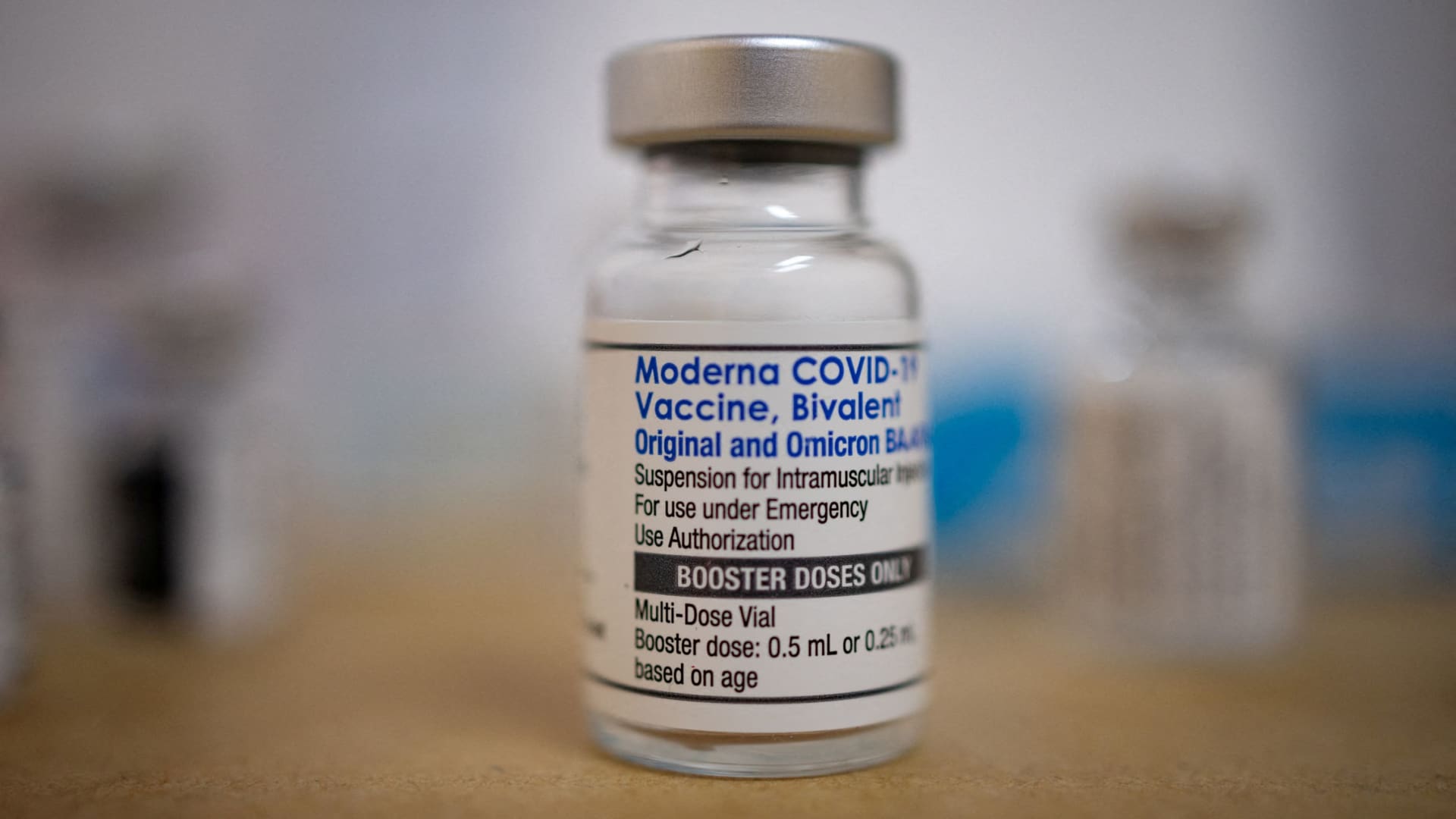
A vial of the Moderna coronavirus booster vaccine targeting BA.4 and BA.5 Omicron subvariants is pictured at Skippack Pharmacy in Schwenksville, Pennsylvania, Sept. 8, 2022.
Hannah Beier | Reuters
The Centers for Disease Control and Prevention on Tuesday recommended that all Americans ages 6 months and older receive updated Covid shots from Pfizer and Moderna, clearing the way for Americans to start receiving the shots within days.
CDC Director Mandy Cohen signed off on a recommendation formed by an independent panel of advisors to the agency earlier Tuesday. The first doses of the new shots will be available at some locations within the next 48 hours, according to the CDC.
"We have more tools than ever to prevent the worst outcomes from COVID-19," Cohen said in a statement. "CDC is now recommending updated COVID-19 vaccination for everyone 6 months and older to better protect you and your loved ones."
Thirteen CDC advisors voted in favor of the "universal" recommendation for Americans during the panel's meeting Tuesday, while one voted against it.
"I think that it's clear that vaccination is going to prevent serious illness and death across all age groups. It is a vaccine-preventable disease," said Dr. Beth Bell, clinical professor at the University of Washington and member of the panel, during the advisory meeting. "And so, for that reason, I favor the universal recommendation."
Dr. Pablo Sanchez, a professor of pediatrics at Ohio State University, voted against the recommendation due to "limited data" on children, infants and other populations, while noting he is "not against this vaccine."
The advisory panel's recommendation comes a day after the Food and Drug Administration approved the two mRNA jabs, which are designed to target the omicron subvariant XBB.1.5. An updated shot from Novavax, which uses protein-based technology, is still under review by the agency.
The FDA approved Pfizer and Moderna's new vaccines for people 12 and older. The agency authorized the shots under emergency use for children 6 months through 11 years old.
The new shots are part of a push by public health officials to update Covid vaccines annually to target more recent strains of the virus — a similar approach to the yearly flu shot. The jab rollout comes as the virus starts to take a stronger hold in the U.S. again.
Hospitalizations have increased for seven straight weeks, and rose more than 15% to 17,418 for the week ended Aug. 26, according to the latest data from the CDC. That number remains below the surge the nation saw in summer 2022.
But the CDC "anticipates further increases" as the U.S. enters respiratory virus season this fall and winter, which is when Covid, respiratory syncytial virus and flu tend to spread at higher levels, CDC epidemiologist Dr. Megan Wallace said during the meeting on Tuesday.
A resident receives a Covid-19 booster shot at a vaccine clinic inside Trinity Evangelic Lutheran Church in Lansdale, Pennsylvania, U.S, on Tuesday, Apr. 5, 2022.
Hannah Beier | Bloomberg | Getty Images
The updated vaccines are expected to help prevent people from getting seriously ill and being hospitalized from Covid infections caused by newer variants.
Pfizer, Moderna and Novavax have all released initial trial data suggesting that their new shots produce robust immune responses against the now-dominant EG.5, or "Eris," variant. That omicron strain is closely related to XBB.1.5 and accounted for 21.5% of all U.S. cases as of Sept. 2, according to the CDC.
Both Pfizer and Moderna also have released initial trial data indicating that their new shots were effective against another omicron variant called BA.2.86. Novavax on Monday said it was still testing its vaccine against BA.2.86. That strain has been detected in small numbers across the U.S., but health officials worldwide are watching it closely due to its high number of mutations.
Moderna's XBB.1.5 vaccine "provides a substantial increase in responses to both the variant in the vaccine, and cross-neutralization of other variants — and this is regardless of prior infection status," said Dr. Fran Priddy, executive director of clinical development for the Covid vaccine program at Moderna, during the advisory meeting Tuesday.
The upcoming arrival of the new vaccines comes months after the end of the U.S. Covid public health emergency.
The end of that declaration means manufacturers will sell their updated shots directly to health-care providers at more than $120 per dose in the private market. Previously, the government purchased vaccines directly from manufacturers at a discount to distribute to all Americans for free.
During the advisory meeting, Moderna said the list price of its vaccine is $129 per dose and Pfizer said the list price of its own shot is $120 per dose. Meanwhile, Novavax said its list price is $130 per dose.
The vast majority of Americans will be able to get the new vaccines at no cost through private insurance or government payers like Medicare.
For the uninsured or underinsured, the Biden administration plans to offer shots for free through its temporary "Bridge Access Program" at health centers, clinics and eventually pharmacies across the U.S. Free vaccines through the program will not be available after December 2024, according to the CDC's website.
The CDC's Vaccines For Children program will provide free Covid shots to children whose families or caretakers can't afford them after the shots move to the commercial market.

 Lynk
Lynk